Online Sport Streaming
exercise rowing machine for home
nfl game pass us
world mens curling live
fences on amazon prime
formula live free
live stream curling world championship
mlb streams free 2022
world hockey game today
motocross online stream
bobsleigh live stream
nascar race now live
nhl toronto maple leafs live
fifa world cup final argentina france
grand prix live
releasesky f1
below deck sailing yacht free stream
stream yankees 2022
athletics live
ice hockey video on demand
remote olympic weightlifting coaching
www livescore com tennis
fences putlocker
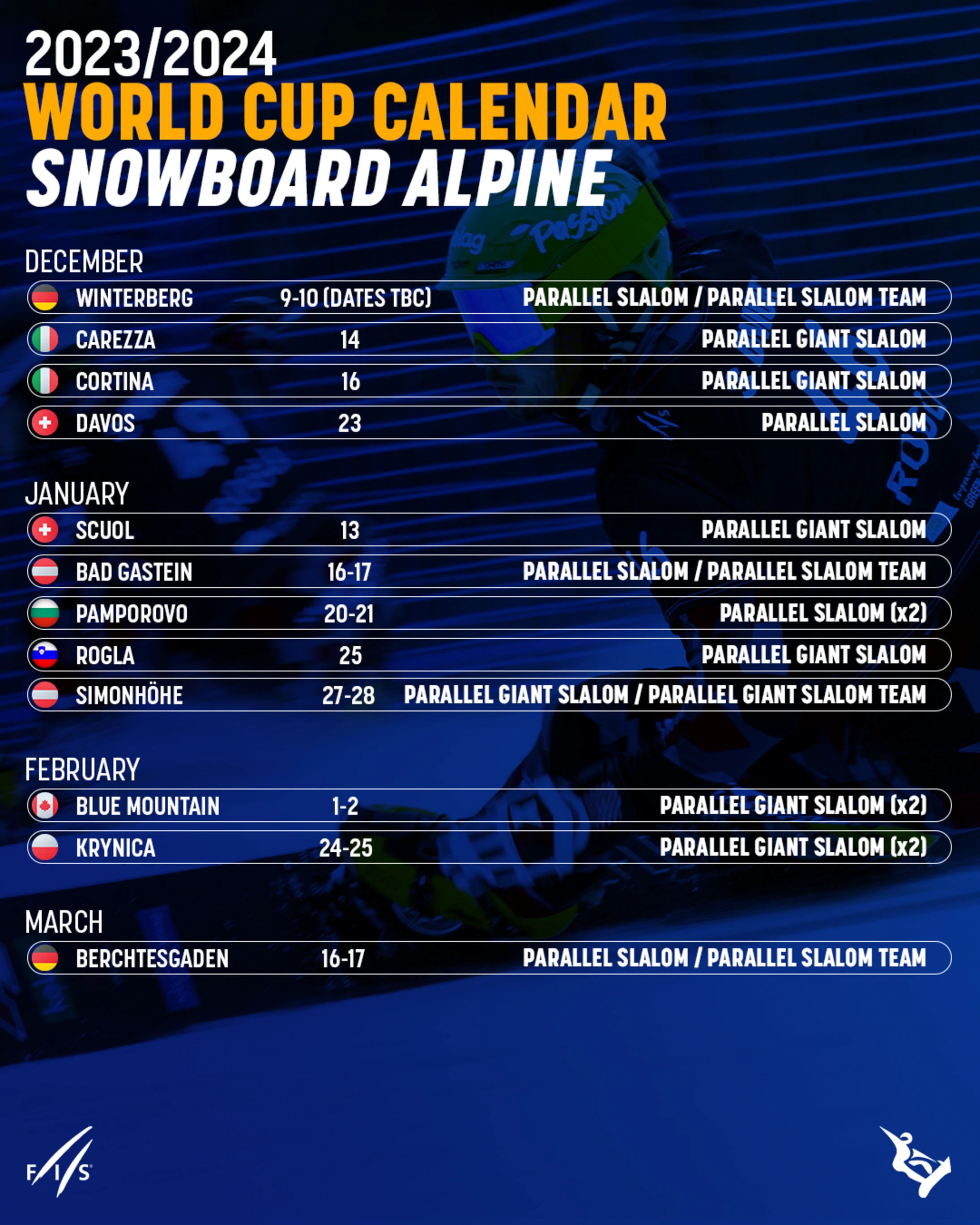
peacock alpine skiing schedule
amazon nfl how to watch
le mans live stream reddit
alpine ski streaming
milwaukee brewers watch
mobile cricket live stream
live world cup cricket 2022
moto2 gp live
womens volleyball on tv
rugby today springboks live streaming
motogp live free streaming 2022
liverowing
ping pong live
stream2watch motogp
online weightlifting coach
medvedev auger aliassime live
judo online
soccer stream sites reddit
horse jumping live
volleyball livestreams
vimeo on demand subscription
below deck sailing yacht stream free
cori gauff live
super bowl 2021 youtube
fis alpine live timing
Explore a winter wonderland and chase legendary status in NBA 2K23. NBA 2K is the ultimate experience for basketball stars in the making, sending you on an immersive journey and bringing your NBA dreams to life. Up Your Game does exactly what its name suggests, assisting you on the court through a series of videos, articles, and more so you can elevate your skills on the court and in popular game modes such as MyCAREER and MyTEAM. Learn from the best in the business when it comes to perfecting your shot, understanding the intricacies of MyTEAM and MyCAREER, and navigating the game as a whole. Watch as he jumps into 2K23 to create his MyTEAM, talks about Seasons, and takes on some fans. Stay up-to-date on the latest NBA 2K23 Patch Updates as they are released for New Gen and Current Gen consoles. Bringing KD into the fold signals a potential golden age of Suns basketball. Despite limited playing time together, Durant and Booker flourished together in the NBA Playoffs and powered the Suns into the second round. In Season 8 of NBA 2K23, close out the year by showcasing your skills in The City (New Gen) and aboard The G.



























/cdn.vox-cdn.com/uploads/chorus_image/image/71195984/FLLN1FoWUAYi94_.0.jpg)










/cloudfront-us-east-1.images.arcpublishing.com/gray/24BDOZTQEVNIFC477O2RYQCBZA.jpg)